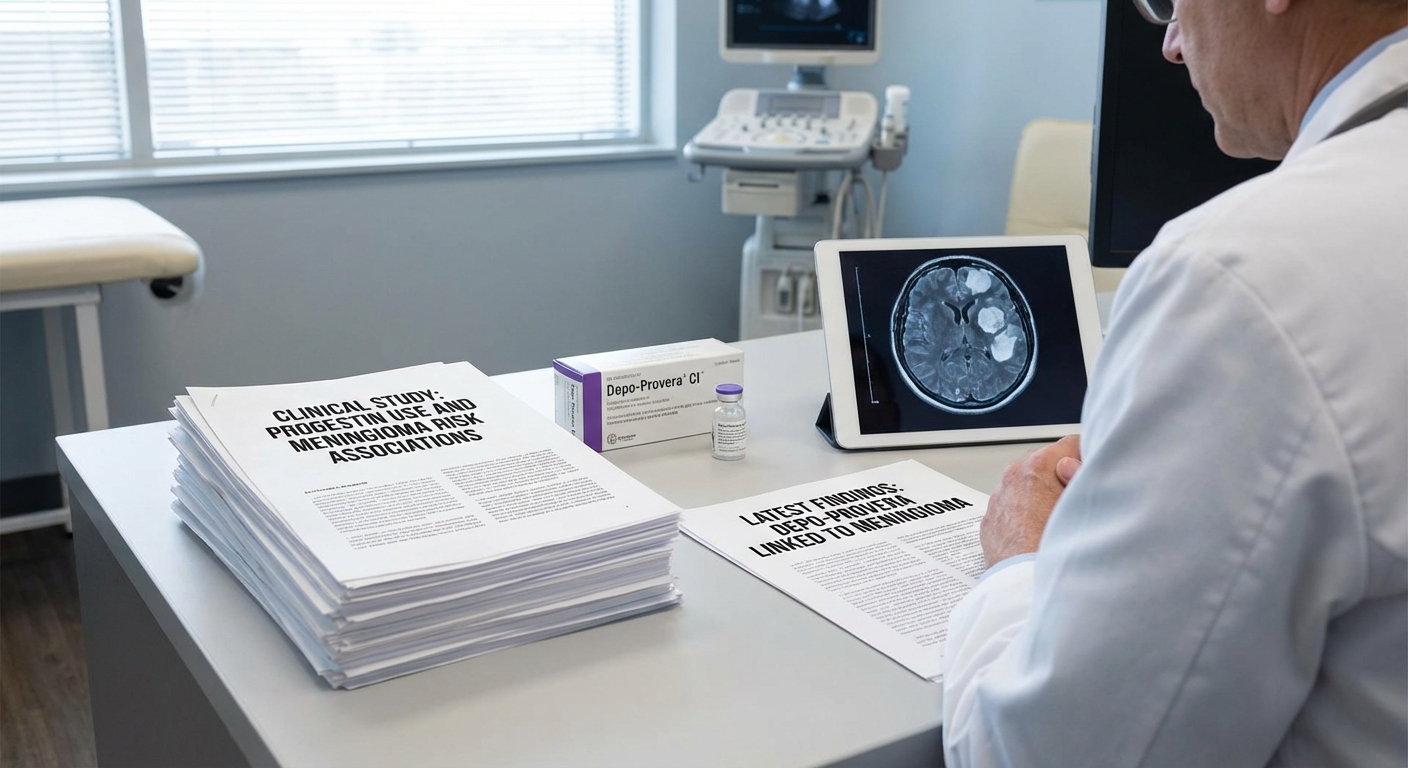
Some women diagnosed with more than one meningioma after Depo-Provera use worry whether the injections contributed to multiple tumors, not just a single growth. This article focuses on what the research and reported cases say about multiple tumors and how lawyers evaluate that history.
If you've used Depo-Provera and have been diagnosed with multiple meningiomas, you may be wondering whether the medication could be responsible. Recent research has revealed a connection between long-term Depo-Provera use and the development of meningiomas, tumors that form on the protective layers surrounding the brain and spinal cord. Understanding whether Depo-Provera can cause multiple brain tumors is important for women who have used the medication and received such a diagnosis.
A landmark French study published in the BMJ found that women who used high-dose progestogen medications like Depo-Provera for extended periods faced significantly elevated risks of developing meningiomas—in some cases, more than five times higher than non-users. While most cases involve single tumors, some women may develop multiple meningiomas, which can present unique challenges for diagnosis and treatment.

Pharmaceutical products and medical research documents
What Are Meningiomas?
Meningiomas are tumors that form on the meninges, the protective layers surrounding the brain and spinal cord. While typically non-cancerous, they can cause serious symptoms as they grow. These tumors develop from cells in the meninges and can vary in size and location, which affects both symptoms and treatment options.
Meningiomas are the most common type of primary brain tumor, accounting for approximately one-third of all brain tumors. While most are benign, they can still cause serious health issues depending on their location and size. Treatment options may include monitoring, surgery, radiation therapy, or a combination of approaches depending on the individual case.
Common symptoms of meningiomas include persistent headaches, vision problems, hearing loss or ringing in ears, memory difficulties, seizures, weakness in limbs, and personality changes. The severity of symptoms often depends on the size and location of the tumor. Some meningiomas may cause no symptoms and are discovered incidentally during imaging for other conditions, while others can lead to significant neurological problems requiring immediate treatment.
Can Depo-Provera Cause Multiple Brain Tumors?
Research on whether Depo-Provera can cause multiple brain tumors is still evolving, but the available evidence suggests that long-term use of the medication may increase the risk of developing multiple meningiomas in some cases. The French study that linked Depo-Provera to meningiomas found that the risk increased with the duration of medication use, and some women in the study were diagnosed with multiple tumors.
Take the first step: a free consultation could clarify your legal options.
Get a free case review and learn whether you may qualify.
Start with a free consultationMultiple meningiomas, also known as meningiomatosis, occur when two or more meningiomas develop simultaneously or sequentially. While less common than single meningiomas, multiple tumors can occur and may be more challenging to treat. The presence of multiple tumors may indicate a stronger association with the underlying cause, such as long-term hormone exposure.
The research suggests that the hormone medroxyprogesterone acetate, the active ingredient in Depo-Provera, may play a role in the development of these tumors. The cumulative exposure from repeated injections over many years may contribute to the development of multiple tumors in some cases. Women who used Depo-Provera for extended periods, particularly ten or more years, may face higher risks of developing multiple meningiomas.
If you've used Depo-Provera for an extended period and have been diagnosed with multiple meningiomas, you may want to learn more about Depo-Provera lawsuit information and your potential rights.

Legal documents and research papers representing ongoing litigation
Understanding Multiple Meningiomas
Multiple meningiomas can present unique challenges compared to single tumors. Understanding how these tumors develop and what they mean for treatment and legal claims is important for women who have received such a diagnosis.
Development Pattern: Multiple meningiomas may develop simultaneously or appear at different times. They can occur in various locations throughout the brain and spinal cord, which can complicate treatment planning. The presence of multiple tumors may indicate a systemic factor, such as long-term hormone exposure, rather than a localized issue.
Diagnosis: Multiple meningiomas are typically diagnosed through MRI or CT scans. The imaging findings can show tumors in different locations, and each tumor may have different characteristics. Accurate diagnosis and mapping of all tumors is important for treatment planning and may be relevant for legal claims.
Treatment Challenges: Treating multiple meningiomas can be more complex than treating a single tumor. Surgery may need to be staged, and radiation therapy may need to target multiple locations. The presence of multiple tumors may affect treatment decisions and outcomes.
Top Tier Legal connects you with experienced mass tort attorneys.
One step: request a free, confidential case evaluation.
Get a free case evaluationPrognosis: The prognosis for multiple meningiomas depends on factors such as the number of tumors, their locations, sizes, and how they respond to treatment. Some cases may have a good prognosis with appropriate treatment, while others may be more challenging.
The Connection Between Depo-Provera and Meningiomas
Recent scientific research has revealed a connection between long-term Depo-Provera use and the development of meningiomas. A landmark French study published in the BMJ found that women who used high-dose progestogen medications like Depo-Provera for extended periods faced significantly elevated risks of developing these tumors.
The study found that women who used Depo-Provera for extended periods, particularly five or more years, faced substantially higher risks of developing meningiomas compared to women who did not use the medication. The risk increased with the duration of use, indicating that cumulative exposure over time plays a significant role in tumor development. Some women in the study were diagnosed with multiple tumors, suggesting that long-term exposure may increase the likelihood of developing more than one meningioma.
The research suggests that the hormone medroxyprogesterone acetate, the active ingredient in Depo-Provera, may play a role in the development of these tumors. While the exact mechanism is still being studied, the evidence indicates a clear association between long-term Depo-Provera use and increased meningioma risk. The presence of multiple tumors in some cases may indicate a stronger association with the medication.
What Is Depo-Provera?
Depo-Provera is an injectable contraceptive medication that contains the hormone medroxyprogesterone acetate. The medication is administered as an injection every three months to prevent pregnancy. It has been used by millions of women worldwide since its approval by the FDA in 1992.
The medication works by preventing ovulation and thickening cervical mucus to block sperm. While effective for birth control, emerging research has raised concerns about potential long-term health risks, particularly related to brain tumor development. Many women have used Depo-Provera for extended periods, sometimes for years or even decades, which may have contributed to the increased risk observed in recent studies.
Depo-Provera injections are typically administered by healthcare providers into the muscle of the arm or buttock. Each injection releases the hormone slowly over time, maintaining contraceptive effectiveness for approximately 12 to 14 weeks. The cumulative exposure from repeated injections over many years may be a factor in the increased meningioma risk observed in long-term users, and may contribute to the development of multiple tumors in some cases.
Free, no-obligation case review. Find out if you qualify.
Free, no-obligation case review. Find out if you qualify.
Check your eligibilityHow Exposure Occurred
Women were exposed to Depo-Provera through regular injections administered by healthcare providers. The medication is typically injected into the muscle of the arm or buttock every 12 to 14 weeks. Each injection releases the hormone slowly over time, maintaining contraceptive effectiveness.
Exposure occurred through regular injections every three months, long-term use over multiple years, cumulative exposure building up over time, and direct injection into muscle tissue. Many women used Depo-Provera for extended periods, sometimes for years or even decades. This long-term, repeated exposure may have contributed to the increased risk of developing meningiomas observed in recent studies, and may increase the likelihood of developing multiple tumors in some women.
The cumulative nature of the exposure is particularly important when considering the development of multiple meningiomas. The hormone builds up in the body over time with each injection, and women who received injections for many years may have had significantly higher cumulative exposure than those who used the medication for shorter periods. This cumulative exposure may explain why some women develop multiple tumors while others develop single tumors.

Evidence and documents in legal proceedings
Who May Be Affected
Women who used Depo-Provera for extended periods may be at increased risk of developing meningiomas, including multiple tumors. Research suggests that the risk increases with the duration of use. Women who used the medication for one to four years may face a moderate increase in risk, while those who used it for five or more years may face significantly higher risks. Women who used it for ten or more years may face the highest risks, including an increased likelihood of developing multiple tumors.
The French study found that women who used high-dose progestogen medications for more than a year had a significantly elevated risk, with some groups facing risks more than five times higher than non-users. Women who have been diagnosed with multiple meningiomas after using Depo-Provera should consider having their situation evaluated to determine whether they may have a valid legal claim.
If you've used Depo-Provera for an extended period and have been diagnosed with multiple meningiomas, you may want to learn more about Depo-Provera lawsuit information and your potential rights.
If you've been affected, a free case review can help you understand your options.
Confidential evaluation at no cost. See if you have options.
Request a free case reviewCurrent Status of Litigation
Litigation related to Depo-Provera and meningiomas is developing as more women learn about the potential connection between their medication use and brain tumor diagnosis. Cases are being filed alleging that manufacturers knew about the risks but failed to adequately warn patients and healthcare providers.
The litigation is in early stages, with cases being filed in various courts. As more scientific evidence emerges and more women come forward, including those with multiple tumors, the litigation may develop into consolidated proceedings similar to other pharmaceutical mass tort cases. Scientific evidence continues to accumulate linking progestogen exposure to meningioma development, and this evidence may support legal claims that manufacturers had an obligation to warn about these risks but failed to do so adequately.
Evidence suggests that the risks associated with progestogen exposure were known for years before adequate warnings were provided. Pharmaceutical companies have a legal obligation to warn patients and healthcare providers about known risks. If you've used Depo-Provera for an extended period and developed single or multiple meningiomas, you may be entitled to compensation for medical expenses, lost wages, pain and suffering, future medical care, and reduced quality of life.
What a Case Review Involves
A case review is an initial evaluation to determine whether you may have a valid legal claim related to Depo-Provera use and meningioma development. This process is confidential, free, and does not create any obligation on your part.
Initial Consultation: The process typically begins with a conversation about your Depo-Provera use history and medical diagnosis. You'll be asked about when you started using Depo-Provera, how long you used it, when you received your meningioma diagnosis, whether you have single or multiple tumors, and what symptoms you've experienced.
Medical Records Review: If you decide to proceed, medical records related to your meningioma diagnosis and treatment may be reviewed. This includes imaging studies that document the presence and characteristics of your tumors. This helps establish the connection between your medication use and your condition. You may also be asked about your Depo-Provera injection history and any documentation you have.
Eligibility Assessment: Based on the information gathered, an assessment will be made about whether you may qualify to pursue a claim. Factors considered include the duration of your Depo-Provera use, the timing of your diagnosis, whether you have single or multiple tumors, and how your case fits within the broader litigation.
Don't wait—statutes of limitations may apply. Get a confidential evaluation.
No obligation. A quick review can clarify your next steps.
See if you qualifyNext Steps: If you may qualify, you'll be informed about the next steps in the legal process. This might include connecting you with an independent law firm that handles Depo-Provera cases. If you don't qualify, you'll receive an explanation of why.
No Upfront Costs: Case reviews are conducted at no cost to you. If you qualify and choose to proceed with a case, most attorneys work on a contingency fee basis, meaning they only receive payment if your case is successful. To understand more about how mass tort cases work, you may find our guide on <a href="/blog/understanding-mass-tort-litigation/">understanding mass tort litigation</a> helpful.
The case review process is designed to be straightforward and respectful of your situation. The goal is to help you understand your options and determine whether pursuing legal action makes sense for your circumstances. If you're ready to begin, you can contact us for a free case review.
Important Considerations
Understanding whether Depo-Provera can cause multiple brain tumors requires recognizing several important points about the process and potential outcomes.
No Guarantees: Legal cases involve uncertainty. Past results in similar cases don't guarantee future outcomes. Each case is evaluated individually based on its unique facts and circumstances. The strength of your case depends on many factors, including the evidence available, whether you have single or multiple tumors, the timing of your diagnosis, and how courts interpret the scientific evidence.
Time Limits Apply: Statutes of limitations set deadlines for filing lawsuits. These deadlines vary by state and depend on factors such as when you discovered your injury and when you were diagnosed. Missing these deadlines can permanently bar you from pursuing compensation, so it's important to act promptly if you believe you may have a claim.
Individual Cases: Each case is handled individually. Your specific circumstances, medical history, medication use details, and whether you have single or multiple tumors will be considered separately. Compensation, if any, will be based on your individual situation rather than a formula applied to all cases.
Scientific Evidence: While research suggests a link between Depo-Provera and meningiomas, including multiple tumors, scientific evidence continues to evolve. Courts will evaluate this evidence, and outcomes may depend on how juries and judges interpret the available research.
Experienced attorneys are ready to review your situation at no cost.
Get a free case review and learn whether you may qualify.
Get your free case reviewLitigation Timeline: Mass tort litigation typically takes several years to resolve. Cases may proceed through discovery, expert witness preparation, and potentially trials or settlement negotiations. The timeline for your specific case depends on many factors beyond your control.
See if you qualify for a free case review to learn more about your potential rights and options. You can also learn more about Depo-Provera lawsuit information and your potential rights.
Important Disclaimers
The question of whether Depo-Provera can cause multiple brain tumors is an important one for women who have used the medication and received such a diagnosis. If you've used Depo-Provera for an extended period and have been diagnosed with single or multiple meningiomas, understanding your legal rights and options is an important first step.
Remember, time is often of the essence due to statutes of limitations. Don't wait to explore your legal options. A free consultation can provide clarity on your situation and help you understand your rights and potential path to compensation. For more information about Depo-Provera and meningiomas, see our articles on meningioma linked to Depo-Provera injections, how long it takes for Depo-Provera brain tumors to develop, MRI findings in Depo-Provera brain tumor cases, symptoms of meningioma after Depo-Provera use, and Depo-Provera lawsuit eligibility. You can also learn more about Depo-Provera lawsuit information and your potential rights.
Top Tier Legal, LLC is not a law firm and does not provide legal advice. This content is for informational purposes only. Submitting information does not create an attorney-client relationship. If you qualify, Top Tier Legal, LLC may connect you with an independent law firm. Past results do not guarantee future outcomes.